Endo Tools Therapeutics Receives FDA 510(k) Clearance for Next-Generation Endomina® EZFuse system
- March 4th, 2026
510(k) clearance enables immediate U.S. commercialization of enhanced soft tissue approximation technology in the gastrointestinal tract
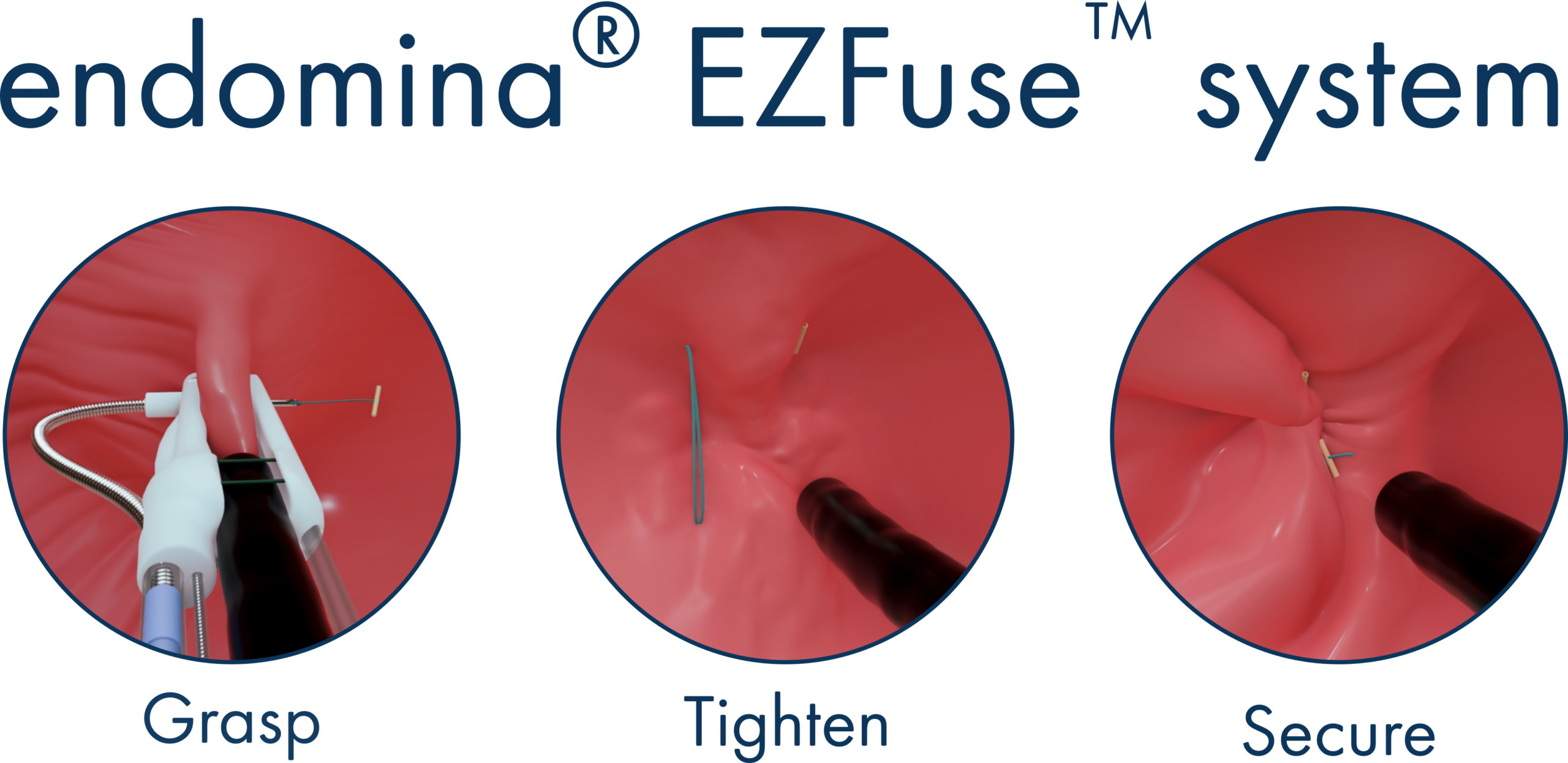
Gosselies, Belgium – March 4, 2026 – Endo Tools Therapeutics (ETT), developers of advanced endoscopic medical devices, today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for its next-generation technology, the endomina® EZFuse system. This advancement in the company’s suturing platform has been engineered with a more intuitive design to deliver faster suturing, greater efficiency, and usability for soft tissue approximation procedures.
“The EZFuse system clearance marks an important milestone in the continued evolution of the endomina system and introduces a number of improvements to simplify workflow and to enhance the physician experience,” said Alexandre Chau, CEO of Endo Tools Therapeutics. “We are excited that the new system is making it easier for physicians to adopt and integrate our device into their practice.”
Early procedural experience with the next generation EZFuse system demonstrates significantly faster suturing time, with a reduction of total procedure time up to 50%, enabling physicians to complete procedures more efficiently without changing their established approach.
The suture’s single-movement mechanism eliminates unnecessary steps, creating a more streamlined workflow. The intuitive design reduces staff and operational burden as well as minimizes user fatigue, helping clinical teams perform procedures with greater ease and predictability. EZFuse suture reduces complexity across the care setting, allowing hospitals and ambulatory surgery centers to optimize resource utilization, improve scheduling flexibility, and expand overall procedural capacity, ultimately leading to improved patient care.
Denis Gestin, Chairman of the Board at Endo Tools Therapeutics, added: “ETT has always been focused on pushing the boundaries of what’s possible in endoscopic innovation. With the FDA clearance of the next generation endomina® EZFuse system, we are reinforcing our commitment to make advanced endoscopic procedures faster, simpler, and more accessible to physicians while improving efficiencies for hospitals and ambulatory centers. This milestone positions the company for accelerated growth and physician adoption in the U.S.”
The company will begin commercialization of the endomina® EZFuse system immediately in the U.S., with plans to broaden availability in key markets worldwide.
About Endo Tools Therapeutics
Located in Gosselies, Belgium, Endo Tools Therapeutics S.A. (ETT) offers a portfolio of advanced, incisionless and minimally invasive endoscopic medical devices. The company’s mission is to develop solutions that expand the range of gastrointestinal procedures that can be performed using standard gastroscopes. By developing minimally invasive treatment solutions, ETT aims to reduce complications and hospital length of stay, while improving patient comfort. In Europe and select Middle East countries, the devices are approved for endoscopic gastroplasty procedures for the treatment of obesity, with over 1,500 procedures already performed, while in the U.S. they are cleared for placement of sutures and approximation of soft tissue in the gastrointestinal tract.
